Table of contents will be automatically generated here...
Definitions
Head trauma can refer to any injury to
the head from a superficial graze, through superficial haematoma to skull
fracture, and life threatening intracranial injury. Traumatic Brain Injury (TBI) refers
more specifically to disruption of the brain tissue by an external mechanical
force. This may lead to a range of
presentations from minimally impaired mental status, with no neurological
deficit, to profound and sustained loss of consciousness.
The Glasgow
Coma scale [1]
has been used to classify severity of traumatic brain injury. This score designates patients as suffering
from mild (GCS 13-15), moderate (GCS 9-12) or severe traumatic brain injury
(GCS <8). Although originally
designed for adults, the Glasgow Coma Scale has been adapted to include
children and infants.[2, 3]. Other criteria used in the various
classifications of head injury severity include presence of post traumatic
amnesia, duration of loss of consciousness, presence of neurological changes,
presence of skull fracture or intracranial lesions.[4]
Introduction
Head
injury in children is common. Traumatic
brain injury is a leading cause of long-term disability and mortality in
children. Children under 3 have the
highest rates of head injury. Most are
trivial or mild head injuries due to falls - around 90% will be discharged from
ED[5].
The majority of head injury in young children is the result of accidental
injury, however in children less than 2 years old, 80% or more of deaths from
head injury are due to non-accidental trauma. [8]
There
is a second peak in head injuries during adolescence. This group has a greater proportion of fatal injuries, where
mechanism is more commonly motor vehicle accident related.[5] In one analysis of children presenting to the
Royal Children's Hospital, Melbourne, 24% of patients
with head injuries had an injury to one or more other body regions and 20 % of all children with severe head injury
had an accompanying cervical spine injury.
Children sustain different patterns of head injury compared to
adults due to their relevant anatomical, physiological and biomechanical
properties. In particular, the following
differences are important[6]:
- The skull is more plastic or deformable in children - the
sutures are open and the bone itself is less rigid than that in an adult
skull. As a result, there is some
movement which absorbs some of the initial impact. The increased plasticity of
the skull may also lead to the production of greater shear forces to vessels
and underlying brain, increasing the frequency of subdural haematomas and
diffuse axonal injury.
- Open sutures may result in the development of “growing
fractures” in infants and children under 3 years old, through the facilitation
of cerebrospinal fluid pulsations along the sutures and fracture lines.
- Open sutures prevent early rise of intracranial pressure - which
may lead to fewer clinical symptoms initially.
- The bones themselves are thinner - and thus offer the brain
inside less protection - and the pattern of fracturing is different to adults
(especially in young).
- They have larger head-to-body weight ratio and weaker neck
musculature. This leads to greater
acceleration / deceleration of the brain and a higher susceptibility to
shearing forces within the brain. These
factors, combined with a relative laxity of the ligamentous structures, also contribute to the higher incidence of
atlanto-occipital dislocation in children (3 times more common than in adults)[7] and a higher rate of ligamentous spinal
injury.
- The combination of a proportionally heavier head and weaker neck
increases neck injury severity including cervical vascular injury leading to
cerebral hypoxia and brainstem dysfunction.
- In addition to the increased head to body weight ratio, children
have an increased cranium to face ratio.
At birth the cranium is about 8 times the size of the face, it
diminishes to 4 times the size of the face by 5yrs of age, and reaches the
adult ratio of 2.5 times the size of the face by adolescence.[6] As a result facial
injuries are less common in small children, and cranial injuries more
common. The relative lack of sinuses in
small children also leads to direct transmission of forces through the face to
the brain.
- Children may lose large volumes of blood due to scalp
lacerations and/or haematomas.
- The immature brain is more prone to injury - containing a higher
proportion of water, and a smaller amount of myelin, than the adult brain. This results in greater deformation following
acceleration-deceleration injury and potentially mechanoporation (where the
transmitted forces open the pores and allow uncontrolled neurotramsitter
release) contributing to the high rates of seizure and cerebral oedema.
- Diffuse
cerebral oedema is a hallmark of paediatric TBI. The exact physiological mechanism
is unknown however may be associated with the fact that children have a higher
cerebral flow than adolescents and adults. [9]
Outcomes from head injuries
depend on both the severity of the initial injury (primary brain injury), as
well as the extent of subsequent complications and how these are managed
(secondary brain injury). A holistic approach to the management of paediatric
head injury therefore includes identification of risks so that primary injuries
can be prevented. This includes maintaining
an increased awareness of inflicted injury, and advocating for the use of
protective devices - such as seat belts, bike helmets or netting around
trampolines which lessen the number of, or decrease the severity of injury
following accidents. This advocacy also identifies and informs parents of those
sports/activities which carry increased risks of head injuries - for example
quad-biking, Aussie rules football, and horse-riding.
Once a primary injury has
occurred, hospital management is directed toward the prevention, identification
and treatment of the secondary injuries.
Pathophysiology
Outcomes from head injuries
depend on the severity of the initial injury (primary injury) and the extent of
subsequent damage from oedema, ischaemia and inflammation (secondary
injury).
The
initial stage of TBI is due to mechanical tissue damage from shearing or
tearing forces on the neurons, glia and blood vessels. In many cases there is direct neural cell
loss and necrotic cell death. The
primary injury triggers secondary biochemical, metabolic and cellular
changes. These include[10]:
-
Excitotoxic
changes – characterized by loss of intracellular glutamate into the
extracellular space from injured neurons. Amongst other processes, this acts to
stimulate NMDA receptors and allow an influx of sodium and calcium ions into
cells which ultimately contributes to cell death through apoptosis and necrosis
- Oxidative
stress – characterized by the accumulation of reactive oxygen and nitrogen species
which lead to cell dysfunction through disruption to the cell membranes, and
induction of an inflammatory response.
-
Mitochondrial
dysfunction – occurs secondary to oxidative stress and large calcium ion influx
and leads to a failure to produce ATP leading to further cell apoptosis.
- Loss of the blood brain
barrier integrity – the primary injury can disrupt the otherwise tight cell
junctions of the blood brain barrier, which allows for an influx of peripheral
immune cells and circulating factors such as albumin, thrombin and
fibrinogen. Through a number of
cascades, this leads to an increase in the osmotic force within the brain,
leading to oedema and higher intracranial pressure.
Primary intracranial injuries
Primary intracranial injuries include:
- Focal
brain contusions - distinct areas of swollen brain tissue - often on the poles
or inferior aspects of the frontal lobes.
Often associated with severe headache, altered level of consciousness
and vomiting.
- Focal brain
laceration – with penetrating injury from either bone fracture edges or foreign
body.
- Diffuse
and focal vascular injuries – resulting in hematomas including:
- Extradural - in adults these are commonly due to arterial
bleeds (especially from the middle meningeal artery). In children, venous bleeding, particularly
from skull fractures, contributes significantly to extradural haematomas -
there may be a delay in signs and symptoms.
- Subdural – Associated with injury to the cortical bridging
veins such as occurs in birth trauma, accidental injury and inflicted head
injury. Additionally subdural haemorrhage is seen with significant adjacent
cortical injury with breach of the cortex and pia and can be seen with
traumatic rupture of arachnoid cysts.
- Subarachnoid - occurs following injury to small vessels
crossing the subarachnoid space.
- Intra-parenchymal – ranging from small foci of blood seen
on targeted imaging to frank haemorrhage and clot formation within the brain
parenchyma such as with avulsed perforating vessels.
- Intra-ventricular - blood in the intraventricular space can
lead to obstruction of CSF flow, and subsequent increase in intra-cranial
pressure.
- Diffuse
and focal axonal injury - this is the result of severe
acceleration-deceleration and rotational forces which lead to stretching and
disruption at a cellular level to axons.
This initial damage leads to a cascade of biochemical and cellular
changes leading to neuronal cell death. Patients
with diffuse axonal injury have an evolving brain injury and are especially
sensitive to secondary injuries. Initial
imaging may be benign or display small areas of punctate haemorrhages.
- Hypoxic
Injury –primary hypoxic brain injury may occur in trauma from seizures or
neural dysfunction with central apnoea. Hypoxic injury is often seen with
inflicted head trauma.
Secondary intracranial injury
This
refers to the injury to brain cells after the original insult. It is a major contributor to overall morbidity
and mortality in traumatic brain injuries - and occurs over hours to weeks
following a primary injury to the brain.
Not all of this secondary brain injury is preventable. Nevertheless, the goals of patient management
are aimed at:
- Optimizing cerebral oxygenation and metabolism
-
Minimizing secondary injury
To
achieve these goals, it is important to understand cerebral haemodynamics.
Cerebral haemodynamics
There are two major principles to consider in order to understand cerebral haemodynamics - the Munro-Kellie hypothesis and autoregulation.
Munro-Kellie Hypothesis
This hypothesis states that the
skull is a rigid compartment filled with essentially non-compressible contents
- the brain, intravascular blood and CSF.
An increase in volume of one of the contents should lead to a decrease
in the volume of another. This can occur
through displacement of CSF to the spinal subarachnoid space, or through
displacement of blood by compression of the venous system. However, the degree of this compensation is
limited, and once it is reached, intra-cranial pressure (ICP) can rise.
Acute
ICP rise can lead to:
- Headache, vomiting, depressed mental status, followed by
posturing, then abnormal vital signs. The
Cushing
reflex describes a reflex bradycardia and systemic hypertension
associated with a raised ICP. It is a late sign of brain injury, and indicates
reduced perfusion of the brainstem, or imminent brainstem / tonsillar
herniation.
- Cerebral herniation - occurs with critical
elevations of ICP. It can occur at a
number of locations in the brain.
- Uncal (transtentorial) herniation - is typically
the result of a unilateral, supratentorial, mass lesion (such as an extradural
haematoma). It arises when the uncus
of the temporal lobe is pushed towards the tentorium, compressing the
midbrain. This leads to pressure on the
3rd cranial nerve - causing ipsilateral pupil dilation initially (via
compression of parasympathetic fibres) and may progress to a complete
oculomotor palsy.
- Central herniation - refers to the movement of
the central brain structures - the diencephalon and parts of the temporal
lobes- through the tentorium
cerebelli. This type of hernia can lead
to tears in the pontine arteries - which is typically fatal.
- Cingulate (subfalcine) herniation - occurs when
the cingulate gyrus is pushed under the falx cerebri.
- Tonsillar herniation - sometimes referred to
as "coning" this refers to movement of the cerebellar tonsils down
through the foramen magnum. It leads to
compression of the lower brainstem - leading to cardiovascular and respiratory
instability.
Autoregulation.
Cerebral perfusion pressure (CPP) is dependent on mean arterial
pressure (MAP) and intracranial pressure (ICP). The uninjured brain is able to
regulate blood flow through a range of blood pressures through autoregulation
of cerebral vascular resistance. Various
conditions such as hypercapnia and acidosis lead to dilation of cerebral
arteries. Alkalosis, hypocapnia and
hypertension lead to cerebral arterial constriction . The injured brain may lose the normal
homeostatic mechanisms for blood flow and when this occurs, blood flow is
dependent on the CPP.
The relationship between ICP and
CPP is shown below:
CPP = MAP - ICP
In injury, a fall in blood
pressure (systemic hypotension) or a rise in ICP will lead to a reduction in
the CPP. A reduction in CPP leads to
cerebral ischaemia (that is a reduction in cerebral blood flow) - which leads
to further neuronal death and cerebral oedema - further increasing ICP and
contributing to a vicious cycle of escalating secondary brain injury.
In
order to prevent cerebral ischaemia, the ICP and MAP must ideally be maintained
within normal limits. In healthy children, a normal ICP is typically less than 10-15
mmHg. In the head injured child, it is
typical to aim to keep ICP below 20mmHg.
The target MAP varies with age - but is typically kept >50-60mmHg. The goal is to achieve a CPP 40-50mmHg with
infants within the lower range and adolescents in the higher range. [14]
Given the concepts outlined above, the general management of
traumatic brain injury focuses on minimising factors that contribute to raised
intracranial pressure, cerebral edema and cerebral ischaemia. This is achieved
by:
-
Avoidance
of hypoxia
- Maintenance
of normocapnia – hypercapnia causes cerebral vasodilation leading to an
increase in cerebral blood flow and worsening of cerebral edema; hypocapnia can
lead to inadequate cerebral blood flow leading to ischaemia.
- Maintenance
of normotension
- Maintenance
of normoglycaemia
- Maintenance
of normothermia - fever can increase cerebral metabolism and increase oxygen
demand.
- Avoidance
of seizures
Initial management of head trauma
Primary survey
In all aspects of trauma management the primary
survey is the first priority. This
should all take less than 1 minute .
A Assess and ensure a patent airway, whilst
maintaining C-spine immobilisation
(see Airway
management)
(see Cervical spine injury)
(see Primary and secondary survey)
B Assess and provide high-flow oxygen; Monitor
oxygen saturation and respiratory rates.
- Assess
indications for intubation – preventing hypoxia is of critical importance for patients with a
severe head injury.
- Aim for pCO2 35-40mmHg
- Aim for pO2 >80mmHg
(see Breathing management)
C Assess adequacy of circulation
- Establish
2 large-bore cannulae.
- Monitor
and document the heart rate and blood pressure.
- Aim for normotension
for age. Permissive hypotension is not indicated in significant head injury.
D Assess level of disability
Maintain
in-line cervical stabilisation at all times
- Rapidly
assess the child’s mental state using the AVPU or GCS scale (see below).
- Assess pupil size,
equality and reactivity.
-
Assess brain stem reflexes and limb reflexes.
- Assess
limb posture: decorticate, decerebrate.
-
Assess focal neurological signs.
- Check
blood glucose level.
AVPU Scale
Use
central stimuli – such as sternal pressure or pinching the trapezius
muscle – to ascertain the child’s response to pain.
- A Alert
- V Responds to
voice
- P Responds to
pain:
- Purposefully – or localises site of pain.
- Non-purposefully.
- Withdrawal/flexor response.
- Extensor response.
- U Unresponsive.
Glasgow
Coma Scale
- The GCS should be scored on the patient’s best responses.
- However, the GCS may be falsely low if any of the following are present:
- Shock, hypoxia, hypothermia, intoxication, post-ictal state or
sedative drug administration.
- Furthermore, the GCS may be impossible to evaluate accurately if the patient
is agitated, uncooperative, dysphasic, intubated or has significant facial or
spinal cord injuries.
- Where a significant head injury has occured, the GCS should be repeated regularly, at least every 15 mins, as deterioration
may occur rapidly.
Glasgow Coma Scale (use if child >4years) and Paediatric Glasgow Coma Scale (use if child is <4 years)| Glasgow Coma Scale (≥ 4 years) | Paediatric Glasgow Coma Scale (< 4 years) |
| | | | |
| Response | Score | Response | Score |
| Eye opening | | Eye opening | |
| Spontaneously | 4 | Spontaneously | 4 |
| To verbal stimuli | 3 | To verbal stimuli | 3 |
| To painful stimuli | 2 | To painful stimuli | 2 |
| No response to pain | 1 | No response to pain | 1 |
| | | | |
| Best verbal response | | Best verbal response | |
| Orientated and converses | 5 | Appropriate words or social smile, fixes, follows | 5 |
| Confused and converses | 4 | Cries but consolable; less than usual words | 4 |
| Inappropriate words | 3 | Persistently irritable | 3 |
| Incomprehensible sounds | 2 | Moans to pain | 2 |
| No response to pain | 1 | No response to pain | 1 |
| | | | |
| Best motor response | | Best motor response | |
| Obeys verbal commands | 6 | Spontaneous or obeys verbal commands | 6 |
| Localises to stimuli | 5 | Localises to stimuli | 5 |
| Withdraws to stimuli | 4 | Withdraws to stimuli | 4 |
| Abnormal flexion to pain (decorticate) | 3 | Abnormal flexion to pain (decorticate) | 3 |
| Abnormal extension to pain (decerebrate) | 2 | Abnormal extension to pain (decerebrate) | 2 |
| No response to pain | 1 | No response to pain | 1 |
The primary survey can help guide initial management steps:
.jpg)
History
Ensure an AMPLE history has been taken (see How children are different). Also ensure to ask about the following:
-
Time of injury
- Mechanism of injury
- Degree of force acting on the head (i.e. speed of vehicle, height
of fall, presence of helmet or restraints)
- Position of patient when found at the scene
- Initial conscious state along with any alteration in
consciousness state (including irritability) and duration
- Seizure
- Apnea
- Nausea / Vomiting
- Headache
- Amnesia
- Past history of bleeding tendency
Secondary survey
Perform a complete secondary survey. In
all cases of head injury, pay particular attention to:
- Checking the head and face for lacerations, bruising, deformity and
tenderness.
- Assessing for signs of base of skull fracture
-
Haemotympanum or bleeding from ear canal
- CSF rhinorrhea or otorrhea
- Periorbital bruising (“raccoon eyes”) – typically bilateral and
associated with anterior cranial fossa injury.
-
Bruising around the mastoid (Battle’s sign) – typically
indicates a fracture of the middle cranial fossa. As with periorbital bruising, Battle’s sign
may present several days post injury.
- Maintaining a high level of suspicion for a cervical spine injury.
- Assessing cranial nerve examination (including visual acuity and
hearing)
- Checking cerebellar function
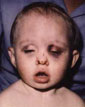
"Racoon eyes"

A Racoon
(see also Primary and secondary survey)
Initial Investigations
Laboratory studies
A general trauma panel is recommended including:
- Venous blood gas
- Full blood count
- Group and Hold
- Coagulation studies
- Electrolytes
- Renal function
- Liver function
- Lipase
- Glucose
Imaging Studies
Skull X-Ray
Infants are more at risk of skull
fractures than older children due to their thinner craniums. The presence or
absence of a skull fracture however, does not always correlate with the severity of
the underlying brain injury.
- A skull X-ray is not performed
routinely in patients presenting with a non-localized head injury, and it is
not used to determine whether a child requires admission. There is no place for
the skull X-Ray “just in case”.
- Skull X-rays generally require more than one plane for
diagnosis and as such low dose helical
CTs are often preferred for greater clinical utility for equivalent radiation
exposure.
CT scan
Performance
of the CT scan should only be undertaken when the patient is stable enough to
be transferred to the CT scanner. They must be fully monitored and accompanied
at all times. Indications for a CT head
include:
- Reduced GCS:
- GCS < or = 8 Requires
urgent CT
-
GCS 9-12 Discuss
timing with neurosurgery; may require urgent
CT if other concerning predictors or clinical signs
are present
- GCS 13-14 May
require urgent CT if other concerning predictors or
clinical signs present (see below).
- Rapid neurological deterioration (> or = 3/2 points) on GCS
Scale.
- Focal neurological signs
- Abnormal pupillary exam
- Abnormal posturing – in particular extensor posturing.
- Full fontanelle and/or separating sutures.
- Bradycardia and hypertension associated with altered conscious
state (Cushing’s reflex is a late sign).
- Requirement for general anaesthesia and a history of prolonged
LOC (> 5 mins)
There
are a number of clinical decision tools to help determine the need for a head
CT in children (PECARN, CATCH, CHALICE). All three tools are highly sensitive,
have external validity and therefore Emergency Departments should consider
instituting one tool into practice.
[13]
Indications for CT after
the phase of initial presentation and assessment: (management directed by
Consultant Neurosurgeon)
- Deteriorating conscious state;
- Unresolving or worsening symptoms of concussion;
- Persistent nausea or vomiting.
Other
important considerations regarding CT and surgery
- If, because of ongoing extracranial bleeding, the patient’s
condition cannot be stabilised with maximal resuscitation, he/she should be
transferred directly to theatre and CT scans postponed.
- The child with GCS < or = 8 should be considered for
ICP-monitoring during surgery. (Discuss with neurosurgeons).
- The child with GCS 9 – 12 or a history of prolonged LOC (> 5
min) requires a CT scan before surgery for other injuries. (Discuss with
neurosurgeons.)
- Non-urgent surgery for other injuries may need to be deferred
for 24 – 72 hours.
Forensic considerations
Whilst it should not interfere with emergency management
documenting history and findings both descriptively and with photography and
preserving clothing or other “evidence” appropriately can help the care of the
child both in elucidating the nature of the mechanism particularly in the case
of inflicted injury and non-verbal children.
Extracranial Injuries
Scalp Injuries
The scalp, being highly vascularized,
can be a source of significant bleeding. In young infants, scalp lacerations,
in particular a subgaleal haematoma, can lead to haemorrhagic shock. [14] All wounds should be explored to check for underlying skull
fractures.
Skull Fractures
Skull
fractures are most common in the first year of life. They may occur in the presence or absence of
external clinical signs - i.e a boggy haematoma, as these may take some time to
develop.
Types of skull fractures include:
- Linear Fractures:
- The most common simple type.
- Seen in the temporal parietal
region.
- Often accompanied by an overlying
haematoma.
- They require no specific
treatment and will heal well.
- A very small minority of infants (<1%) may develop a “growing
skull fracture” (see below).
- Diastatic
Fractures:
- These are caused by traumatic separation of the sutures most
commonly lamboidal.
- Growing Fractures:
- These are seen in infants where there has been associated dural
injury. With the brain pulsation and rapid growth in this age the fracture widens
“grows” with herniation of injured brain through the torn dura up in to the
fracture site. These fractures generally present sometime after the initial
injury, usually as a persistent swelling or pulsatile mass and requires surgical repair.
This is screened for in at risk age groups by a clinical check of fracture
healing by palpation 6-8 weeks post injury.
- Depressed
Fractures:
- Usually due to a direct blow.
- May be easily overlooked.
- All local wounds should be
palpated.
- Need discussion with a neurosurgical team and may require
surgical intervention.
- Open
Fractures:
- Should be explored and closed.
- Should have vigorous wound-toileting and irrigation because of
the risk of infection.
- Patients should be covered with broad-spectrum antibiotics.
- Basal
skull Fractures:
- These may be difficult to see radiologically, although the
clinical suspicion should be high if “battle’s sign” / “raccoon eyes” or a CSF
leak is present
- Do not use
nasal airways or nasogastric tubes in these patients due to the risk of
intracranial placement.
- There is a risk of meningitis with streptococcus pneumoniae, but
antibiotics are not given empirically.
It
is important to evaluate children with skull fractures thoroughly and a CT scan
should be included as part of the assessment. Most children should be routinely
admitted for a period of observation. Where a child has injured their
head, always remember to consider the potential for an associated cervical
spine injury. Assess the cervical spine clinically and radiologically where
indicated. (see
cervical spine assessment)
Management of extracranial injuries
CT scan is typically indicated in focal injuries. Discuss with
neurosurgical team.
- Admission to a neurosurgical centre is required in most cases
- All wounds should be explored to check for underlying skull
fractures.
- To prevent wound infection, all patients with external
compound head injuries should receive antibiotics. In Victoria, RCH recommends:
-
IV flucloxacillin – which is suitable for most wounds
- The addition of gentamicin and metronidazole if contamination is
present.
- An appropriate sterile dressing should cover the wound.
- Prophylactic antibiotics are not indicated for patients with internal
compound fractures (that is - base of skull) with CSF leak.
- All of these patients should be closely observed, and discussed
with a neurosurgical center who will usually admit them to ensure the leak has
resolved.
- Those patients who develop a fever with CSF leak, or have a
history thereof, require emergency neurosurgical consultation and empirical
antibiotics such as flucloxacillin and cefotaxime.
Intracranial injuries
Mild traumatic brain injuries / concussion (GCS 13-15)
Mild TBI and
concussion are interchangeable terms. Sports concussion is a subtype of mild
TBI. According to
The WHO Collaborating Centre Task Force on Mild Traumatic Brain Injury, mild
TBI is defined as:
- One or more
of the following:
- confusion
or disorientation
loss
of consciousness for 30 minutes or less
- post-traumatic
amnesia for less than 24 hours
- other
transient neurological abnormalities such as focal signs, seizure, and
intracranial lesion not requiring surgery;
- Glasgow Coma Scale score of 13–15 after
30 minutes post-injury or later upon presentation for healthcare.
The Task Force also
states the manifestations cannot be due to a penetrating head injury. [15] Others have
restricted mild TBI to those with GCS 14-15 as patients with GCS 13 have a risk
of intracranial lesion like those with moderate TBI. [16] In general,
mild TBI does not result in in head CT or MRI changes. This finding however is
not universally agreed upon and may change as new neuroimaging modalities are
introduced.
Management of mild TBI:
The greatest
risk to patients with apparent mild
TBI is the delayed onset of an intracranial haematoma or brain swelling. Both
are the most common causes of avoidable death in this subset of patients. The
decision to perform a head CT or not in a child with apparent mild TBI is therefore
critical. There
are a number of clinical decision tools, with high sensitivity and external
validity, to help determine the need for a head CT in children (PECARN, CATCH, CHALICE[KA1] /NICE).[13]
(see Radiology). According to the 2014 National Institute for Health and Care
Excellence (NICE) Guidelines (UK), factors that would prompt a head CT in
children include: [17]
Any one of:
-
Suspicion of non-accidental injury
- Post traumatic seizure where there is no history of
epilepsy
- If > 1 year old, any GCS of <14 on initial
assessment or any GCS <15 at 2hrs following injury
- If <1 year old, any GCS <15 on initial
assessment (or subsequently)
- If < 1 year old any bruise, swelling or laceration
larger than 5cm on the head
- Any suspected open or depressed skull fracture / tense
fontanelle
- Any sign of basal skull fracture (haemotympanum,
‘panda’ eyes (that is periorbital ecchymosis/bruising - more commonly referred
to as “raccoon eyes” in Australia), CSF leakage from the
ear
or nose, Battle’s sign)
- Focal neurological deficit
Or, in the
absence of any of the above, two or more of:
- Witnessed loss of consciousness for more than 5
minutes
- Abnormal drowsiness
- Three or more discrete episodes of vomiting
- Dangerous mechanism (high speed road traffic accident
either as pedestrian, cyclist or vehicle occupant, fall from a height of
greater than three metres, high-speed injury from a projectile or other
object).
- Amnesia (antegrade or retrograde) lasting more than 5
minutes
Observation, in
hospital, for a minimum of four hours is advised where there is there is only
one of the latter set of conditions. Home
observation is another option for patients with normal mental status, normal
neurological exam and the availability of a reliable adult parent/carer.[17] Of
note, it can be difficult to have a reliable neurological exam in infants and a
higher degree of suspicion is required.
Prognosis of mild TBI:
Typical course
is resolution of confusion within 24 hours. Concussion results in an
inflammatory state of the brain which is then vulnerable to second injury
(second impact syndrome). Clear guidelines on rest, recovery,
return to activity and return to risk activities must be given at discharge. Post-concussion
symptoms are common. These include:
- Physical
symptoms – nausea, dizziness, headache, blurred vision, sleep disturbance
- Cognitive
deficits – Poor memory, attention and executive function
- Emotional
changes – depression, irritability, anxiety and emotional lability
- Post-concussive
symptoms gradually resolve during the following 12 weeks.
Some patients
with mild TBI have persistent symptoms or develop neuropsychological deficits
however it is difficult to predict in which patient group this occurs. A
pre-morbid neuropsychiatric disorder is strongly related to persistence of
symptoms for 3 months or longer after injury. Patients with persistent symptoms
for greater than 3 months may benefit from treatment guided by teams
specialized in mild TBI/concussion. [15]
Sports-related concussion: Post-concussion
symptoms typically resolve within 7-10 days after a first concussion. [15] Patients should
be counselled to have a gradual return to sport.
Recommendations to families post
discharge:
See ‘Head
Injury Handout’. (end of this chapter)
Moderate
traumatic brain injury (GCS 9-12)
Clinical
features can include:
- Brief loss of consciousness at time of injury – with or without
amnesia to the injury.
- Alert on examination, or responds to voice. May be drowsy.
- Two or more episodes of vomiting.
- Persistent headache.
- Up to one single brief (less than 2 min) convulsion, occurring
immediately after the impact.
- May have a large scalp bruise, haematoma or laceration. These
can be responsible for significant reduction in the circulating blood volume.
The area should also be palpated to help assess for an underlying depressed skull
fracture.
- Normal examination otherwise.
Management
of moderate TBI:
Consult
Neurosurgical team and consider CT scan if:
- A
persistent headache, large haematoma or possible penetrating wound may need
further investigation.
-
At 4 – 6 hours, the child is still drowsy or vomiting.
- There is any deterioration during this time.
-
Admit
to a ward knowledgeable in paediatric TBI for observation.
Most patients will initially rapidly
recover from the effects of concussion within a 12-24
hour period. A delay or reversal of recovery suggests haemorrhage, cerebral oedema, or
infection.
Severe traumatic brain injury (GCS <8)
Clinical features can include:
- Prolonged loss of consciousness at time of injury.
- Increasingly severe headache, with or without vomiting
- Decreased conscious state – responsive only to pain, or
unresponsive.
-
Abnormal/change in behaviour.
- Bleeding/CSF leak from nose or ears.
- Localising neurological signs (unequal pupils, lateralising
motor weakness).
- Signs of increased intracranial pressure:
- Cushing response of bradycardia with hypertension: a late sign in children which occurs
secondary to tonsillar herniation / decreased brainstem perfusion
- Changes in pupillary size or reactiveness : uncal herniation
leads to a dilated non-reactive pupil due to compression of the ipsilateral
oculomotor nerve by the ipsilateral uncus (usually results from unilateral
haemorrhage or collection).
- Penetrating head injury. All scalp wounds and lacerations should
be explored, to check the underlying skull is intact prior to suturing.
- Seizures (other than single brief [less than 2 min] convulsion)
occurring immediately after the impact.
- Out of hospital arrest – inflicted head injury in babies and
infants may present with unexplained arrest / loss of consciousness in the
absence of external signs of trauma.
Management of severe TBI
Consult
neurosurgery and intensive care early. Manage
as a multi-trauma patient.
Goals
of treatment are:
- Optimizing cerebral oxygenation and metabolism
- Minimizing secondary injury
- Rapidly identify intracranial injuries requiring surgical intervention
Specific
management
Optimise oxygenation
- Maintain a patent airway
- Maintain adequate
ventilation and oxygenation.
- Target PO280mmHg
- Target PCO2 35-40 mmHg
Optimise cerebral perfusion
- Optimise head position
- Keep head of bed at 30°
- Midline head position
- Ensure normotension for age
- Correct hypovolaemia
- If unable to maintain
blood pressure with fluid resuscitation, may consider vasopressor
(noradrenaline or vasopressin) in discussion with intensive care
- Use of sedation and analgesia to prevent any movement,
coughing, pain that can increase ICP
- Give additional sedation prior to suctioning
- Consider muscle relaxation, in discussion with neurosurgery
Other strategies to minimise secondary injury
- Give dextrose containing maintenance fluids to maintain normoglycaemia (4-8 mmol/L)
- Maintain
normal serum sodium
- Ensure temperature control
- Maintain normothermia (36°C)
- Seizure management (see
below)
- Elevated ICP management (see below)
Imaging - Arrange urgent head CT.
Management of post traumatic seizures
Post
traumatic seizures are a common event following severe brain injuries. The incidence in this group (i.e. GCS <8) has
been reported as between 19-39%. However,
it is worth noting this risk increases further in those who are young
(<2yrs), have a subdural haematoma, and those who have an inflicted
injury.[18] The
aggressive management and prevention of seizures in the head injured patient is
required to prevent secondary brain injury.
The mechanisms by which seizures worsen secondary brain injury
include:[19]
- Increasing
metabolic demand
- Increasing
CBF and ICP
- Increasing
neurotransmitter release
- Increasing
relative hypoxia / ischaemia
Traditionally,
post- traumatic seizures have been categorized into either:
- early
post-traumatic seizures – which occur within 7 days of the initial injury
- late
post-traumatic seizures – which more than 7 days after the initial injury
However, it has been suggested
that “early post-traumatic seizures” group can be further subdivided into the
following groups[19]:
- Impact
seizures – occurs at the time or within minutes of injury. Is typically a brief, generalized seizure
associated with rapid and complete recovery.
- Immediate
post traumatic seizure – occur within the first 24hours post injury. Most early
post traumatic seizures occur within this time period.
- Delayed
early seizures – occurring between days 2 and 7. Seizures occurring in this time frame are more
likely to be associated with severe brain injury such as intracranial
haemorrhage.
Risk
factors for early post traumatic seizures include[19]:
- Age
<2
- Abusive
or inflicted head injury
- Intracranial
haemorrhage
- Severe
TBI (i.e GCS <8)
- Prolonged
loss of consciousness
- Depressed
or open skull fracture
Aggressive
treatment of seizures is important to prevent secondary brain injury. Management includes the following:
-
Support
airway and breathing, apply oxygen by mask, monitor, note time of onset.
- Secure
IV access, check bedside serum glucose and send urgent specimen for calcium /
electrolytes and venous blood gas. Treat hypoglycaemia.
- Give
benzodiazepine immediately – IM or IO if IV access is
unavailable. Consider a reduced dose to
avoid hypotension (for example in the multiply injured patient, where there is also a degree of haemorrhagic shock) and hypoxia.
- Repeat
benzodiazepine after 5 minutes of continuing seizures.
- If seizure
continues for a further 5 – 10 minutes, commence IV anti-epileptic drug. Current recommendations favour the use of IV
phenytoin, however, leviteracetam should
be considered where there is concern over the patient’s haemodynamic status as
it is less likely to cause hypotension.
- Seek
senior assistance if seizure not controlled – involve PICU / PIPER early.
-
Anticipate
need to support respiration.
-
Anticipate
hypotension induced by anti-epileptics.
- Manage
with fluid resuscitation and vasopressors as needed.
- For
severe TBI, prophylactic phenytoin
may be considered to reduce the incidence of early posttraumatic seizures. [20]
Any early post traumatic seizure, apart
from a brief (<2min) impact seizure, warrants investigation with imaging (CT
or MRI brain).
|
Drug
|
Route
|
Dose
|
Comments
|
|
Midazolam
|
IV/IO/IM
|
0.15mg/kg
|
Give immediately in the seizing head injured patient. Caution is required in the injured, hypotensive
pt who may not tolerate a full dose of midazolam. Avoid the intra-nasal route in children
with any awareness during seizures due to irritation
|
|
IN
|
0.2-0.5 mg/kg (max 10mg)
|
|
Buccal
|
0.3mg/kg (max 10mg)
|
|
Phenytoin
|
IV / IO
|
20mg/kg
|
Avoid if there is concern about toxicological cause being associated
with or leading to the patient experiencing a head injury. May also precipitate a degree of
hypotension – either anticipate and prevent this or consider using
leviteracetam
|
|
Leviteracetam
|
IV/IO
|
20-40mg/kg
|
Consider as second line agent where 2 doses of
benzodiazepams have failed to control seizures
|
|
Propofol
|
IV/IO
|
Titrated to effect
|
Use only with involvement of senior staff confident with
airway management. Beware of hypotension.
|
|
Thiopentone
|
IV/IO
|
Titrated to effect
|
Use only with involvement of senior staff confident with
airway management. Beware of hypotension.
|
Management of elevated ICP
Detection
of elevated ICP requires invasive monitoring – however, an acute rise in ICP
can be inferred from certain clinical symptoms (described in the cerebral
haemodynamics section above).
Manage in consultation with
neurosurgery and intensive care.
Priority
is to minimize ICP while awaiting surgical evaluation. In
addition to the management strategies discussed above, additional therapies
include:
First
tier therapies
- Sedation, analgesia and neuromuscular blockade
- Ensure deeply
sedated
- Use
neuromuscular blockade if not already given
- C-spine immobilisation
- If deeply
sedated and/or muscle relaxed, may consider the removal of the cervical collar if it thought to be impairing venous return. However, the use sandbags to stabilise the head is recommended and it is imperative to ensure all staff are aware that C-spine is not clinically cleared.
- Hyperosmolar therapy - typically hypertonic
saline (3%), (although mannitol is widely used, due to lack of evidence, the most recent Guidelines for the Acute Medical Management of Severe Traumatic Brain Injury in Infants, Children and Adolescents can only recommend the use of hypertonic saline [12]).
- Mechanism of effect:
- Initial rapid and
transient (< 75 minutes) reduction in blood viscosity leading to viscosity-mediated
vasoconstriction (if intact autoregulation) and a reduction in cerebral blood
volume without a decrease in blood flow. A decrease in cerebral blood volume decreases
ICP.
- Slower onset
osmotic effect (onset 15-30 minutes; lasts up to 6 hours) in which there is
movement of water from the brain parenchyma into the systemic circulation.
Requires intact blood-brain barrier.
- Possible side effects: Renal injury, natriuresis
Second tier therapies
- Temperature control
- Prevent
hyperthermia
- Can consider mild-moderate hypothermia, in isolated head injuries, in discussion
with neurosurgery and paediatric intensive care. However, in the multiply injured patient, hypothermia may
exacerbate any existing coagulopathy.
- Barbiturates
- Decrease ICP
by decreasing cerebral metabolic rate
- Thiopentone 1
mg/kg
- Possible side
effects: Hypotension
Medical management may not
be able to relieve elevated ICP secondary to a large haematoma or severe
cerebral oedema. Emergent surgery may be
required, so early consultation with neurosurgery is necessary.
Localised brain injury
Injuries
caused by a direct blow and confined to a focal area of the head. These
injuries may cause:
- Simple or compound depressed skull fractures -these are common.
- Lacerations and compound wounds - which may become infected.
- Focal contusions or laceration of the brain may be present to a
varying size or depth.
- Concussion may be absent
Management
Initial management is resuscitation, as for severe head injury (see
skull fractures above).
- CT scan may be required. Discuss with neurosurgical team
-
Admission to a neurosurgical centre is required in most cases
- All wounds should be explored to check for underlying skull
fractures (see above).
Intracranial haemorrhage
Intracranial haemorrhages will
cause a rise in the ICP with the clinical features discussed above. It is essential
that a neurosurgical consultation occurs as soon as possible, for advice on acute
management and for definitive treatments, such as a surgical evacuation.
Subdural haematoma
This is the most common type of intracranial bleed. It may occur at any age but often seen in infants. Subdural haematomas:
-
Are venous in origin, caused by tearing of the bridging meningeal
veins.
- May be associated with significant underlying parenchymal damage.
- May present with seizure.
- Are management with urgent surgical review and prompt surgical
intervention, when required.
- May be secondary to inflicted head trauma - hence, it is essential to consider this whether there are any red flags regarding inflicted injury for these patients - especially in infants.
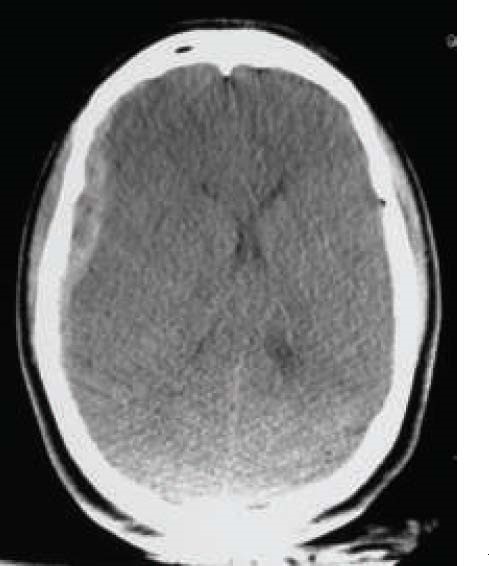 Subdural haematoma
Subdural haematoma
The CT scan above shows a subdural haematoma in the right hemisphere, associated with mass effect as evidenced by midline shift.
Extradural haematoma
Extradural haematomas are often associated with a skull fracture. The haematoma forms a mass and the patient may have an initial
lucid period before a collapse. Typical, extradural haematomas:
- are a unilateral lens shaped collection (may also be
confined to the internal surface of the suture at the dural attachment) of
acute blood or mixed acute and hyperacute.
- present with headache, vomiting, and altered consciousness. Seizures are relatively rare.
Management requires urgent neurosurgical intervention with surgical evacuation of the haematoma and repair of any vessels.
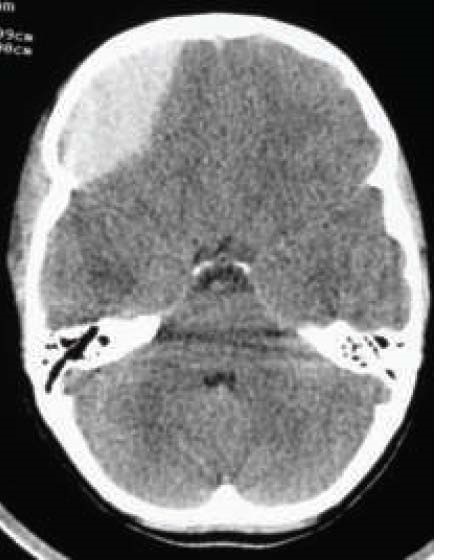 Extradural haematoma
Extradural haematoma
The CT scan above shows an extradural haematoma overlying the right frontal lobe.
Subarachnoid haemorrhage
Subarachnoid haemorrhages are common in trauma. They arise as a consequence of tearing of the small vessels of the pia mater.
The blood distributes within the subarachnoid space, rarely causing a mass
effect and it may remain relatively focal. Subarachnoid haemorrhages are:
-
Usually an indicator of trauma rather than a lesion requiring
treatment in itself.
- Should be discussed with the neurosurgical team.
Cerebral contusions
These occur from high-energy
impact, when the cerebral cortex impacts against the sides of the fixed skull
vault. The injuries occur at two points:
- At the site of the traumatic impact (“coup injuries”)
-
And at the opposite point of impact (“contracoup injuries”)
They
are most likely to occur where the brain is less cushioned, i.e less CSF
- Under the surface of the frontal poles
-
At the poles of the temporal lobes
Effects of cerebral contusions:
-
The patient may demonstrate generalized head injury signs or may
have focal neurological deficit/behavioural/ mental state disturbances.
- Seizures may occur and worsen secondary brain injury.
-
Contusions frequently “blossom” following an injury and as such
usually require admission for neurological observation and potentially repeated
imaging.
RCH head injury handout
RCH has two head injury handouts that can be given to caregivers and older children. The first covers general advice, and the second provides advice on graduated return to school and sport
References and further reading
- Teasdale, G. and B. Jennett, Assessment of coma and impaired consciousness. A practical scale.
Lancet, 1974. 2(7872): p. 81-4.
- Reilly, P., et al., Assessing the conscious level in infants and
young children: a paediatric version of the Glasgow Coma Scale. Child's
Nervous System, 1988. 4(1): p.
30-33.
- Borgialli, D.A., et al., Performance of the Pediatric Glasgow Coma
Scale Score in the Evaluation of Children With Blunt Head Trauma. Acad
Emerg Med, 2016. 23(8): p. 878-84.
- Thurman, D.J., The Epidemiology of Traumatic Brain Injury in Children and Youths A
Review of Research Since 1990. Journal of child neurology, 2016. 31(1): p. 20-27.
- Crowe, L., et al., The epidemiology of paediatric head injuries: data from a referral
centre in Victoria, Australia. Journal of paediatrics and child health,
2009. 45(6): p. 346-350.
- Pinto, P.S., et al., The unique features of traumatic brain
injury in children. Review of the characteristics of the pediatric skull and
brain, mechanisms of trauma, patterns of injury, complications and their
imaging findings--part 1. J Neuroimaging, 2012. 22(2): p. e1-e17.
- Hall, G.C., et al., Atlanto-occipital dislocation. World
Journal of Orthopedics, 2015. 6(2):
p. 236-243.
- Bruce, D.A. and Zimmerman, R. A. Shaken Impact Syndrome. Pediatr Ann,
1989. 18(8): p. 482-4, 486-9, 492-4.
- Fuhrman, B.P.
and Zimmerman, J.J. et al., editors. Pediatric Critical Care. 5th
ed. Philadelphia: Elsevier; c2017. Chapter 60, Structure, function, and development
of the nervous system; p. 826.
- Lozano, D et al. (2015) Neuroinflammatory responses to
traumatic brain injury: etiology, clinical consequences, and therapeutic
opportunities. Neuropsychiatr Dis Treat
11:97-106
- Nichols, D.G.,
editor. Roger's Textbook of Pediatric Intensive Care. 4th ed.
Philadelphia: Lippincott Williams and Wilkins; c2008. Chapter 56, Head and
spinal cord injury.
- Adelson, P.D.
et al., Guidelines for the acute medical
management of severe traumatic brain injury in infants, children, and
adolescents. Chapter 5. Indications for intracranial pressure monitoring in
pediatric patients with severe traumatic brain injury. Pediatr Crit Care
Med, 2003 July. 4(3 Suppl):S1-82.
- Babl, F.E. et al., Accuracy of PECARN, CATCH, and CHALICE head injury decision rules in
children: a prospective cohort study. Lancet, 2017 Apr 11 [Epub ahead of
print].
- Baren, J.M. et al., editor. Pediatric
Emergency Medicine. Philadelphia: Saunders/Elsevier; c2008. Chapter 17, Head
Trauma.
- Servadei
F, Teasdale G, Merry G, and the Neurotraumatology Committee of the World Federation
of Neurosurgical Societies, Defining
acute mild head injury in adults: a proposal based on prognostic factors,
diagnosis, and management. J
Neurotrauma 2001. 18: p.
657–64.
- Levin, H. S., Diaz-Arrastia, R. R, Diagnosis,
prognosis, and clinical management of mild traumatic brain injury. Lancet Neurol, 2015. 14(5): p. 506-517.
- NICE
clinical guideline 176 Head injury:
assessment and early management available at: https://www.nice.org.uk/guidance/cg176
- Bennett,
KS et al. Seizures in Children with
Severe traumatic Brain Injury. Pediatr Crit Care Med. 2017 18(1):54-63
- Arndt,
DH et al. Early Posttraumatic Seizures in the Pediatric Population. Journal of Child Neurology 2016 31(1):46-56
- Kochanek
P.M. et al., Guidelines for the acute
medical management of severe traumatic brain injury in infants, children, and
adolescents--second edition.Pediatr Crit Care Med. 2012 Jan. 13 Suppl 1:
p.S1-82.