Introduction
Aim
Definition of Terms
Assessment
Special Considerations
Companion Documents
Links
Evidence Table
Introduction
A medication administered into a muscle is known as an intramuscular (IM) injection. The IM route allows for rapid absorption of specific medications. Choosing a muscle is dependent on the medication volume and the age or size of the patient. Poor technique and incorrect landmarking of
the injection site can lead to site reactions, sub-optimal medication absorption and adverse events.
For IM immunisation information, please refer to
The Australian Immunisation Handbook.
Aim
This guideline aims to facilitate the administration of intramuscular injections to maximise the therapeutic effect while minimising potential complications and patient discomfort.
Definition of terms
Anterolateral: Anterior and lateral position situated in the front and to the side.
Greater
trochanter:A bony prominence at the top of the femur where the hip and thigh muscles are fixed.
Trauma
informed practice: ‘Trauma-informed care is a way of approaching interactions with children and families in which providers remain cognizant of the impacts of trauma while also taking actions to prevent potentially traumatic experiences’ (Boles, 2017).
Z- tracking: A technique used to prevent medication leakage, particularly for oily injections. Displace the skin and subcutaneous tissue by pulling the skin laterally or downward from the injection site. Holding it taut, quickly and smoothly insert the needle into the muscle at a 90-degree
angle. This technique is preferred in adult/ adolescent patients.
Assessment
Clinical judgement is required when selecting an injection site and needle length.
Summary
| Age |
Site |
Volume |
Needle Size |
Special Considerations |
|
Neonate or Infant
(
<18 months) |
Vastus Lateralis
(Recommended site for all IM Adrenaline) |
1-3mls |
25g x 25mm or 23g x 25mm
(16mm length can be used for neonates or small infants
<2 months) |
Preferred site in neonates and children under walking age but can be used in any age group (up to 5mls can be given in adults) |
|
Infant or child
(18 months–3 years) |
Vastus Lateralis or Deltoid |
1-2mls |
25g x 25mm or 23g x 25mm |
Clinical judgement is used based on the size of the child and their development (see considerations above and below) |
|
Child or Adolescent
(3–11 years) |
Deltoid |
1-2mls |
25g x 25mm or 23g x 25mm |
Can be given in children >12 months if muscle mass is developed |
|
Infant, child or adolescent
(>7 months) |
Ventrogluteal |
Up to 3mls |
23g x 25mm or 21g x 38mm
(38mm length preferred in larger, overweight or obese patients) |
Only recommended at The Royal Children's Hospital (RCH) if appropriately trained
|
| Infant, child or adolescent |
Dorsogluteal |
Up to 4mls |
23g x 25mm or 21g x 38mm
(38mm length preferred in larger, overweight or obese patients) |
|
When IM has been prescribed
Site
selection and land marking
Vastus Lateralis
The anterolateral aspect of the thigh, or vastus lateralis muscle, is the preferred IM site for neonates and infants. Up to 1ml can be injected into this site in neonates, up to 3mls in children and up to 5mls in adults. This is the preferred site for anaphylaxis
management in all ages.
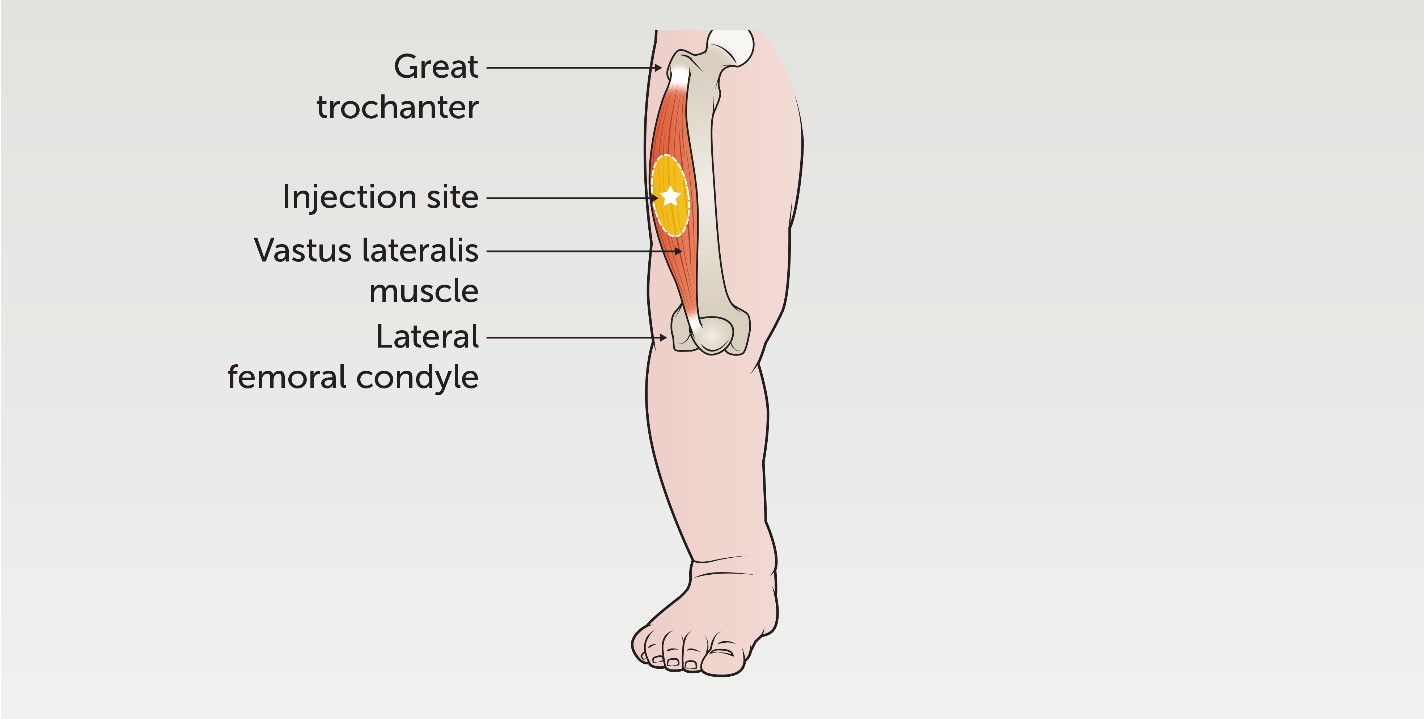
To landmark the vastus lateralis, position the patient lying down or being held by a parent. Palpate the muscle below the greater trochanter and above the lateral femoral condyle (knee joint). Divide the muscle into thirds and administer the injection into the middle third of the muscle, in the outer anterolateral
aspect, lateral to the midpoint of the thigh.
Deltoid
The deltoid muscle is the preferred injection site in children aged 3-18 years when muscle mass is more developed. It is suitable for small volume injections. The recommended volume is 1ml; however, up to 2mls can be administered.
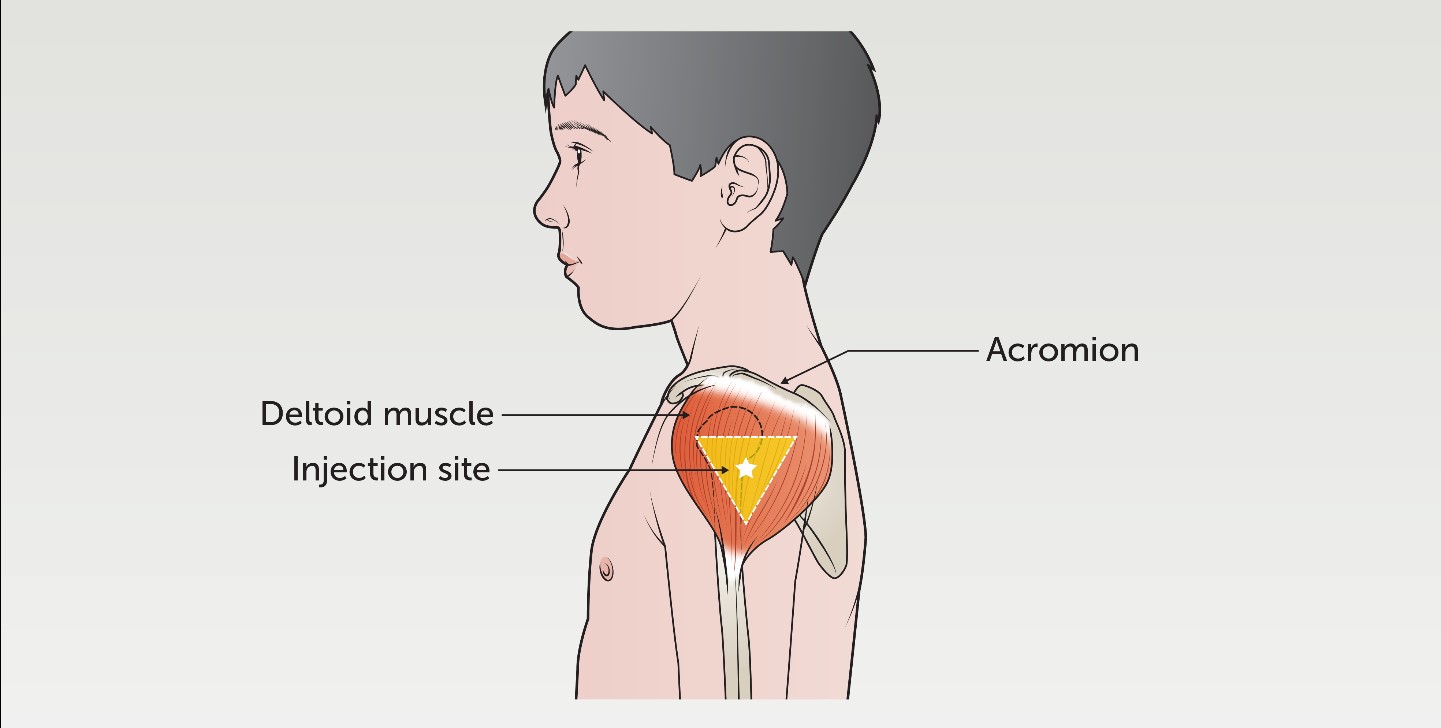
The deltoid muscle is a rounded triangle shape. To landmark this site, the patient should be sitting comfortably with their arm visible from the shoulder to the top of the elbow. Palpate the acromion (outer edge of the scapula) and trace an imaginary inverted triangle below the
shoulder. The injection should be given 3-5cms below the acromion, in the middle of the triangle.
Dorsogluteal
The dorsogluteal is not the preferred site due to its proximity to the sciatic nerve and major blood vessels. If landmarked correctly however, it is safe to use. Up to 4mls can be injected into this site.
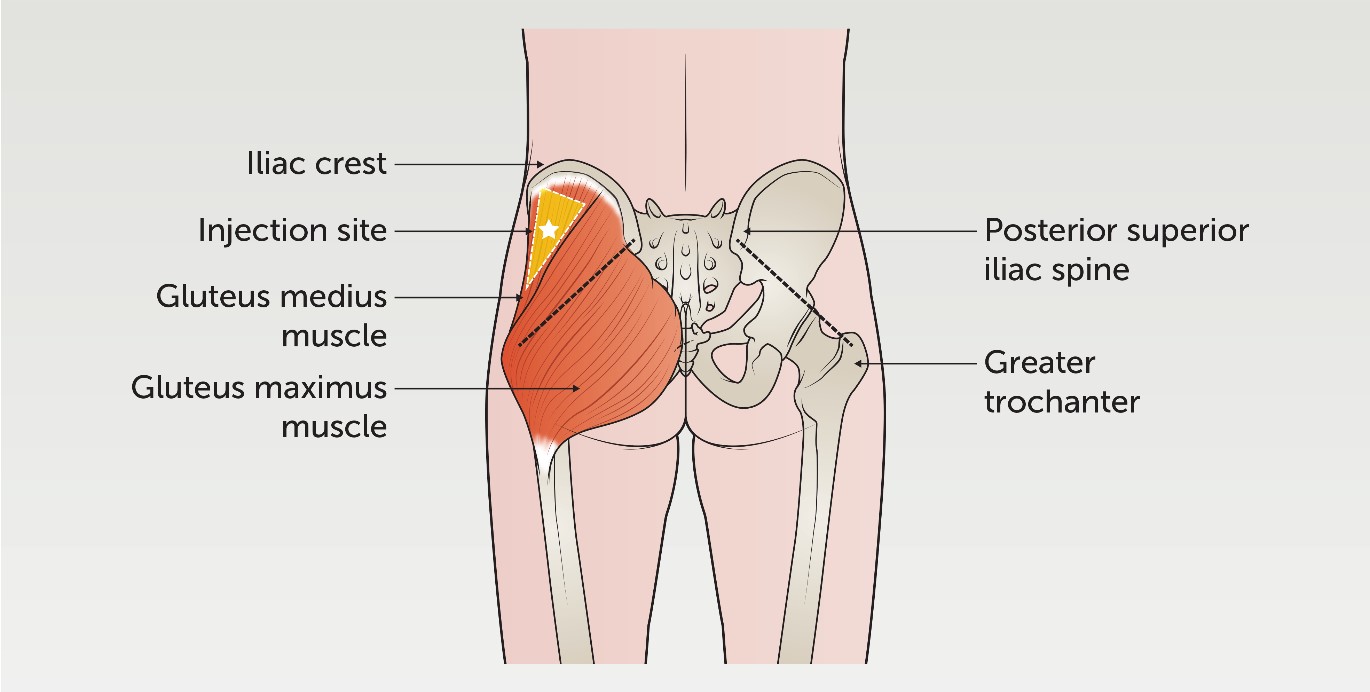
The patient can be positioned lying on their stomach or standing up. The muscle is located in the upper outer quadrant of the buttock, approximately 5-8cm below the iliac crest. For confirmation, an imaginary line can be drawn between the posterior superior
iliac spine and the greater trochanter of the femur.
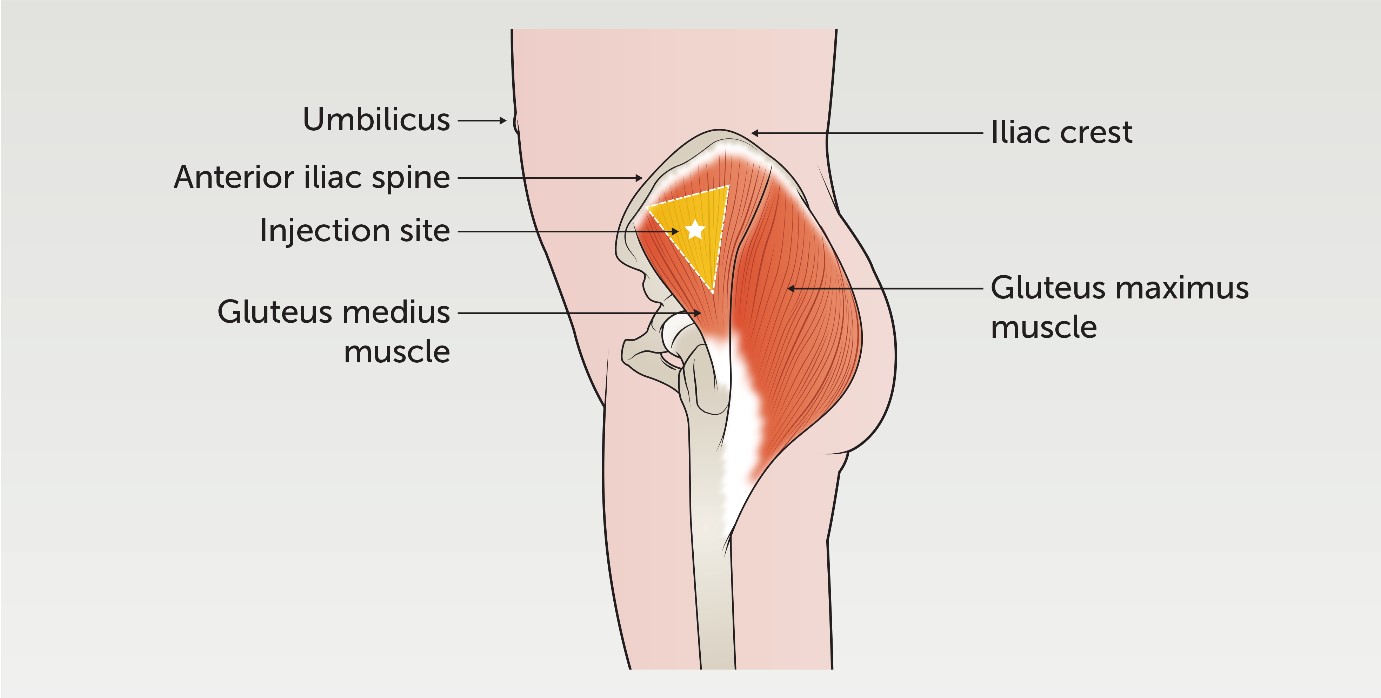
The patient can be positioned lying on their stomach, side or standing up. To locate the muscle, place the palm of a hand over the greater trochanter of the femur, facing the index finger and thumb towards the umbilicus, along the anterior iliac spine. Place the middle finger toward the iliac crest creating a
‘V’ shape. The injection is given in the middle of the ‘V’.
Needle size
A hypodermic needle is used to administer IM injections. These come in different sizes (gauge and length) and are selected based on the patient’s size and the muscle used. A higher gauge needle refers to a smaller outside diameter of the needle tubing.
Needle sizes most commonly used at the RCH include:
- 25g x 25mm ORANGE
- 23g x 25mm BLUE
- 21g x 38mm GREEN
Equipment
- Appropriate size needle for administration
- Drawing up needle and syringe (if medication not pre-filled)
- Sharps container
- Alcohol impregnated swab (if area visibly soiled)
- Cotton ball
- Band-Aid (check for allergies)
- Personal Protective Equipment (PPE) for hazardous medications or infectious patients
The procedure
- Prepare patient and obtain consent
- Perform the five moments of hand hygiene
- Prepare equipment as per
Aseptic Technique
- Complete the six rights of medication administration
- Don PPE if required (particularly for hazardous medications)
- Draw up or prime medication as per Paediatric
Injectable Guidelines
- Position patient in a safe and comfortable position
Procedure Management Guideline
- Consider the use of comfort techniques such as distraction, buzzy bee, ice or a countdown
- Landmark injection site (see above)
- Clean site with an alcohol swab (if required)
- Stretch the skin flat (Z-tracking if applicable)
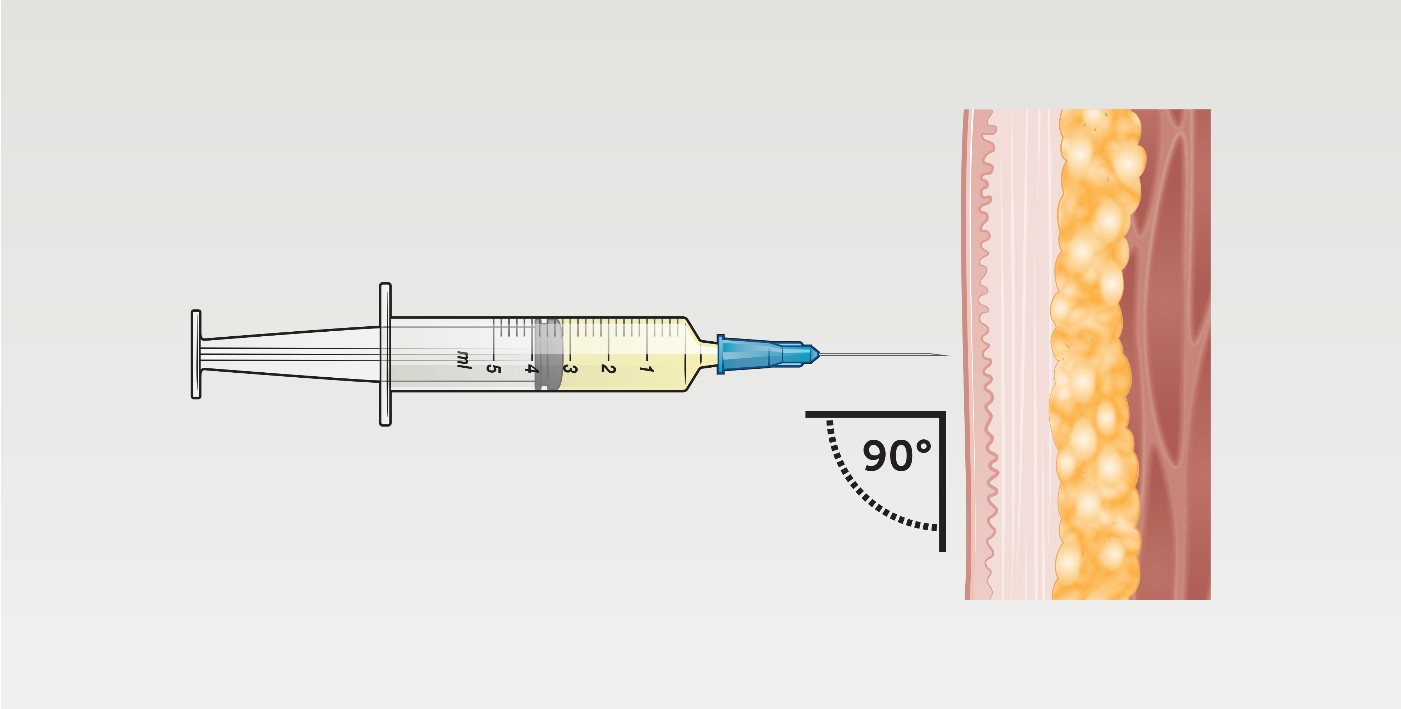
- Inject the needle to the hub at a 90-degree angle
- Do not aspirate or drawback as this can increase pain and discomfort in children
- Inject the medication at a slow and steady pace
- Remove the needle and apply a cotton ball
- Place the needle in a sharps container
- Apply Band-Aid
- Remove PPE (if required)
- Perform the five moments of hand hygiene
Special considerations
- Monitor for immediate adverse reactions, e.g., fever, rash, vomiting, shortness of breath. See
Kids Health Info: Allergic and anaphylactic reactions for more information
- Advise patients to remain in the hospital/ on the ward for 15–20 minutes following their injection. The patient may require longer observation periods depending on the medication they receive
- Provide education to the patient and their family on signs and symptoms to monitor for, e.g., pain, redness, abscess, bruising at the site or anaphylaxis
- Warm or cold compresses can be applied to the area for comfort
Companion documents
Links
The Australian Immunisation Handbook (health.gov.au)
Evidence table
The evidence table for this guideline can be accessed here.
Acknowledgements
All images and videos were created by the Royal Children’s Hospital Creative Studios for the purpose of this guideline.
Please remember to
read the disclaimer.
The development of this nursing guideline was coordinated by Mica Schneider, RN Specialist Clinics, and approved by the Nursing Clinical Effectiveness Committee. First published May 2022.