Introduction
Safe insulin therapy in the inpatient setting is paramount for all patients requiring insulin therapy. Patients in Diabetic Ketoacidosis (DKA) or other acute reasons may require intravenous (IV) insulin infusion therapy. An IV infusion of Regular short-acting insulin
– Actrapid HM™ or Humulin R Regular™ – will be used in critically ill patients who experience changes in insulin requirements as their clinical condition changes; see DKA Clinical Guidelines –
https://www.rch.org.au/clinicalguide/guideline_index/Diabetes_Mellitus/.
Transitions between IV and S/C insulin therapy are a critical time for patients, requiring a considerate and deliberate approach to avoid glycaemic excursions and ensure a safe transition.
The transition from IV to S/C insulin will be initiated by the Endocrinology and Diabetes medical team supported by unit-based nursing staff – therefore all nursing staff are required to understand the transition process required to allow a safe and successful transition to S/C insulin.
Knowing the type of insulin that has been used in IV infusion, and insulins to be used for the S/C injection is vital. IV insulin infusions are made up with Regular short-acting insulin, such as ACTRAPID HM™/Humulin R Regular™ (refer to Insulinssection of this Nursing Clinical Guideline) which is different to the insulins used
for transition to S/C insulin. There is a combination of rapid-acting insulin - NovoRapid®/Humalog® and a basal acting insulin - Optisulin® (glargine) used for the transition to S/C insulin. Rapid-acting insulin often used will be NovoRapid® and the long-acting insulin will be Optisulin® (glargine).
Regular short-acting insulin Actrapid HM™ is used in an IV as it is a human neutral insulin and has a half-life around 5 – 7 minutes. This allows for infusion rates to be readily adjusted to achieve glycaemic targets according to the biochemical assessment of the patient in DKA; see DKA Clinical Guidelines -
https://www.rch.org.au/clinicalguide/guideline_index/Diabetes_Mellitus/.
Aim
Provide guidance to nursing and medical staff in the transition of IV insulin to S/C insulin regimen to ensure:
- The transition process/guideline is streamlined to allow transition to occur when medically safe and ready, and monitored to ensure the transition has been successful –
- Parents, carers and/or patients are actively included and educated throughout the transition to S/C insulin.
Definition of Terms
Diabetic Ketoacidosis (DKA): the patient will present or display signs of hyperglycaemia (high blood glucose), metabolic acidosis (low pH), and blood ketone (high blood ketone).
The biochemical criteria for diagnosis of DKA are:
- Serum glucose > 11 mmol/L
- Venous pH
< 7.3 or Bicarbonate < 15 mmol/L
- Presence of ketonaemia/ketonuria
Refer to:
https://www.rch.org.au/clinicalguide/guideline_index/Diabetic_Ketoacidosis/
Assessment prior to transition
- Patient is alert and orientated
- Biochemical readiness has been medically assessed:
- Able to tolerate fluid and feeling hungry
The patient must be medically ready to safely transition to S/C insulin regimen – see DKA Clinical Guideline -
https://www.rch.org.au/clinicalguide/guideline_index/Diabetes_mellitus/
The
biochemical criteria supporting transition to S/C insulin are:
- pH level: > 7.3
- HCO3 (bicarbonate)
level: > 15 mmol/L
- Provide guidance to nursing and medical staff in the transition of IV insulin to S/C insulin regimen – as RCH unit-based schedules may influence the approach – timing of mealtimes, or access to meals/food, or time of transition evening as opposed to
daytime
- Transition process/guideline is streamlined to allow transition to occur when medically safe and ready, and monitored to ensure the transition has been successful – to avoid workflow challenges and potential patient risks for S/C insulin transition
- Transition to S/C insulin regimen for parents, carers, or the patient to commence education or self-care of their diabetes with unit nursing staff, or diabetes educators
- Based on initial assessment of patient, newly diagnosed with Type 1 Diabetes, and their affect to medical intervention and procedures, please consider and prepare patient for their first S/C insulin injection. Please refer to the following link:
https://www.rch.org.au/
Clinical Guidelines (Nursing) : Procedure Management Guideline (rch.org.au)
Consider the risks of transitioning, which include:
- Rebound/Severe
Hyperglycaemia if there is more than 3 – 4 hours between rapid-acting insulin doses, or no bridge with basal insulin. This may lead to the generation or rise of blood ketones and a need to restart IV insulin.
- Hypoglycaemiaas a result of stacking basal insulin given at the time of transition and again at the standard administration time
- Insulin access:
- Imprest of insulin: location of medication stock/s, Unit based Pharmacists are best source for MAR orders to ensure stock available on any unit.
Management for transitioning to S/C insulin
Night before
anticipated transition to S/C insulin
Ensure medical readiness of patient:
- Diet order placed in EMR (Electronic Medical Record) to ensure a meal for morning transition is ready
- Confirm MAR order for insulins is ordered and
insulins on unit – ready for morning transition
- Ensure order for referral to Diabetes Educator and Dietitian Inpatient Referral is placed if newly diagnosed or liaise with Endocrinology and Diabetes medical team to see if they want referral activated
- Ensure family member or patient is aware of
transition, as they may need to commence administrating insulin in the morning; supported and supervised by nursing staff
Morning of
transition S/C insulin regimen
Ensure patient has remained within medical readiness parameters:
- Confirm patient within normal biochemical
parameters (if unsure contact Endocrinology and Diabetes team)
- obtain a blood gas to assess pH and bicarbonate levels – order should be in EMR
- Ensure clear MAR insulin orders have been placed in EMR – if uncertain contact Endocrinology and Diabetes Medical team to discuss
- Ensure all insulins are available or
equipment to be used on unit
- Ensure diet has been ordered and coming to
unit
- Check whether a Diabetes Educator Inpatient Referralhas been made – if so notify them of time of transition to S/C insulin on the unit
S/C insulin transition: immediately prior to S/C injections
- Ensure insulins dose/s are in the MAR - both a Rapid-acting insulin (Novorapid HM™) and Basal insulin (Optisulin™ glargine or Levemir®)*
- Ensure insulin/s are available on unit
- Ensure meal is available for patient and the patient remains able to eat
- Perform blood glucose (point of care testing – POCT) and ketone – record in EMR result
- Ensure meal ready and available on unit
Administration of S/C
insulin/s and ceasing of IV insulin infusion
- Administer ordered S/C insulin – nursing staff can support or supervise if appropriate - a parent, carer or patient to self-administer their insulin
- Patient must eat no later than 15 minutes after S/C insulin administration
- Turn off IV insulin infusion and associated IV fluids 30 minutes after S/C insulin administration
- POCT blood glucose and ketone check 2 hours after IV infusion have been ceased
*Note: Special considerations
with Glargine and Levemir doses:
Glargine is usually given in the evening. The first dose of basal insulin, usually Glargine, is often given at the same time as the first Rapid acting insulin dose, as the insulin infusion will be ceased and therefore no background insulin will be present.
Often ½ the dose of Glargine calculated by the Endocrinology and Diabetes medical team will be given in the morning and the other ½ or another dose given in the evening. Clarify with Endocrinology and Diabetes Medical team if unsure about Glargine order in MAR.
Levemir can also be used as a first basal insulin to be given in the morning of transition. Later in the evening the Glargine dose can be given. This allows basal insulin coverage during the day.
Initial Assessment and Nursing Care of S/C insulin
transition -
- Monitor POCT blood glucose and ketone – perform before meals (pre-prandial) and aim for between 4.0 – 7.0 mmol/L prior to meal. It might take several days of insulin adjustments made by the Endocrinology and Diabetes medical team to achieve this – there will be reportable blood glucose and ketone levels to contact the medical
team
- Patient remains alert and orientated
- Patient tolerated meal
Ongoing nursing assessment and diabetes care
- POCT* glucose checks – pre-prandial before breakfast (1st meal), lunch, dinner, bedtime (usually 2200) and 02:00 overnight
- Patient will be required to have 3 main meals (breakfast, lunch and dinner) with morning or afternoon snack/s – meals must be adequate and contain the quantities as discussed with dietitian. Meals are provided by the hospital or unit kitchen, and we should not encourage takeaway
meals or snacks whilst an inpatient
*Nursing staff should encourage the patient, or parents or carers to use their own home device provided by DNE. They may have a Libre 2 sensor and the sensor glucose data this does not replace finger lancing blood glucose or ketone checking – however encourage them to scan device and then perform blood glucose
or ketone check too.
S/C Insulin – transition to ‘Basal
Bolus’ insulin regimen (Mutiple Daily Injections – MDI)
A combination of Rapid-acting insulin (Novorapid®/Humalog®) and long-acting insulin (Glargine) will be ordered on the MAR. Novorapid®/Humalog® prior to main meals, and Optisulin® once a day (on first day this may be given as a split dose for 1st 24 hours after IV insulin infusion
ceased).
Specifics about Insulin orders
- Ensure the insulin doses are noted in the MAR at the beginning of the shift so there will be no delays with insulin administration especially around mealtimes.
- S/C insulin can be administered either by disposable pen devices or non-disposable pen/s (usually only started with Diabetes Educator). On RCH imprest both insulin pens and insulin cartridges should be in stock on the units who manage or are transitioning a
patient to S/C insulin.
- Some insulin orders will be for half-unit requests such as 2.5 units. Unless the Diabetes Educator has provided the device (NovoPen ECHO® or JuniorSTAR®), insulin cannot be given in half-unit increments as these pen devices are not readily available on the units. Contact the diabetes medical team if an order
for 0.5 units has been made in the MAR and there is no half-unit pen available as often the dose can be rounded up or down depending on the clinician’s review.
- Specific RCH diabetes links about insulin management:
https://www.rch.org.au/diabetes/type-1-diabetes-toolkit/About_insulin/
Insulin information
Types of insulin available at RCH - specific to orders in
the MAR
The Pharmacists associated with the unit are the best resource to accessing and ensuring insulin is available on your unit for patients use and when ordered on the MAR.
Refer to Appendix 2 – Imprest
of Insulins at RCH – details of unit location of insulins and pharmacy imprest of insulins.
Most used for S/C
basal bolus regimen*
Rapid-acting insulin – NovoRapid HM™
(Aspart) or Humalog®(Lispro)
- Administered 15 minutes prior to meals
- Sites are those easily accessed by patient – abdomen, outer thigh and if parents performing outer arm – sites must be rotated
- Onset after administration 15 mins – if possible, have the patient wait 15 minutes after administration before eating
- Used on units and for discharge with patient for day-to-day diabetes management
- Can be used for correction of blood glucose excursions in-between mealtime, if necessary, often with presence of blood ketone > 1.0 or reportable medical orders
Long-acting insulin – Optisulin® (Glargine)
- Given once a day (as inpatient usually give before bed)
- Site best tolerated is the hip region
- No peak – steady state reached by 4 – 6 hours and lasts for 24 hours
- First day transition to S/C insulin, ½ dose may be given in morning and the rest later in evening
Other insulins
Short-acting insulin – Actrapid HM™
(Regular) – usually only available in vial
- Used only for insulin infusions – available in emergency or ICU units, and would need to be requested to unit
- When used as S/C insulin action onset 30 mins, peak 2-4 hours and duration 6 – 8 hours
- In an infusion it action is immediate, and must be stopped 30 minutes after first S/C Rapid-acting insulin administration
- Half-life is 5 – 7 minutes and generally only used with insulin infusions
Long-acting insulin – Levemir® (Detemir)
- Used for only patients requiring BD (twice daily insulin) insulin regimens or occasionally with basal bolus regimens at clinicians’ request
- Given with morning and evening rapid- or short-acting insulin
- Insulin action – onset 3 – 4 hours, peak 9 hours and duration 12 – 24 hours
*Please review in: Appendix 1 – Profile of insulins and
Insulins action for more detailed information about insulins
Appendix
Appendix 1 Profile of Insulin and Insulin Action
Profile of Insulins
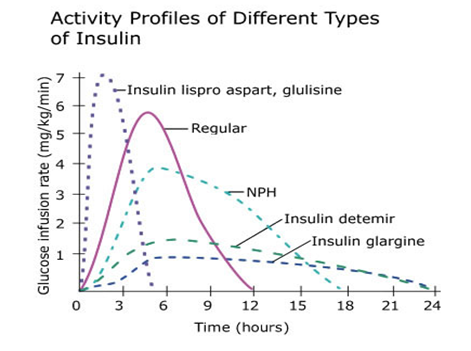
Insulin Action
| Type of Insulin |
Onset |
Peak |
Duration |
Appearance |
| Fast-acting |
|
|
|
|
|
Short-acting – Regular insulin:
Actrapid HM™
Humulin R Regular™ |
½-1 hr. |
2-4 hr. |
6-8 hr. |
clear |
|
Rapid-acting insulin:
NovoRapid®
Humalog® |
< 15 min. |
1-2 hr. |
4-6 hr. |
clear |
| Long-acting |
|
|
|
|
| Levemir® |
1 hr. |
5 hr. |
12+ hr. |
clear |
| Optisulin®glargine |
1 hr. |
Flat, Max effect in 5 hrs. |
24+ hr. |
clear |
https://dtc.ucsf.edu/types-of-diabetes/type2/treatment-of-type-2-diabetes/medications-and-therapies/type-2-insulin-rx/types-of-insulin/
Appendix 2 - Imprest of Insulins at RCH Coming soon
Links
RCH CPG
Family info
RCH Pharmacy
RCH Departmental Guideline
Evidence Table
The evidence table for this guideline
can be viewed here.
Please remember to read the
disclaimer.
The revision of this nursing guideline was coordinated by Rebecca Gebert, CNC, Department of Endocrinology and Diabetes, and approved by the Nursing Clinical Effectiveness Committee. Updated February 2023.