Note: This guideline is currently under review.
Introduction
Aim
Definition of Terms
Assessment
Management
Insertion and Securing of NPT
Ongoing Assessment and Monitoring
NPT Trial Out
Other Considerations
Companion Documents
Links
Evidence Table
Introduction
Pierre Robin Sequence (PRS) occurs in approximately 1 in 6,000 babies in Victoria. It is a congenital condition comprised of a triad of characteristics including micrognathia, glossoptosis, and upper airway obstruction. Upper airway obstruction is a result of micrognathia and glossoptosis. A cleft palate is a common finding in babies with PRS, however a recent international consensus was that micrognathia, glossoptosis and upper airway obstruction should be the defining features in diagnosing PRS, and a cleft palate is not considered a definitive feature. Given that the airway obstruction occurs at the base of the tongue, the main treatment aim is moving the tongue forward to alleviate the obstruction. The use of a nasopharyngeal tube has proven to be a highly effective method in alleviating upper airway obstruction in babies with Pierre Robin Sequence.
Aim
The aim of this guideline is to provide medical and nursing staff with a framework for the insertion and management of a modified NPT in neonates with PRS, to relieve upper airway obstruction.
Definition of Terms
- Modified Nasopharyngeal Tube (NPT): At RCH, a modified nasopharyngeal tube is created using an endotracheal tube (ETT). The appropriate sized ETT is cut down to recommended length to make a nasopharyngeal tube that is inserted to facilitate airway patency. The modified NPT is designed to provide relief of upper airway obstruction in a minimally invasive manner, whilst enabling oral feeding. An alternate term used in literature is nasopharyngeal airway. A modified NPT is specific to this guideline and used in some babies with PRS on the Butterfly ward.
- Glossoptosis: The posterior displacement of the tongue into the oropharynx and hypopharynx resulting in upper airway obstruction.
- Micrognathia: A retro positioned and hypoplastic mandible, causing the tongue to fall back into the oropharynx resulting in upper airway obstruction.
Upper Airway Obstruction: Management of upper airway obstruction plays a critical role in the treatment of PRS. Airway obstruction is mostly due to glossoptosis, whereby the prolapsed tongue occludes the airway, resulting in difficulty breathing. Upper airway obstruction results in a failure of airflow into the lungs, despite adequate inspiratory effort. Increasing respiratory effort can worsen the obstruction, as increased intra-thoracic pressure collapses the soft tissue structures inwards. Airway obstruction may be intermittent and may also take time to develop. Some infants with PRS may not present for days to weeks after birth.
- Cardio Respiratory monitoring: Technological measurement of heart rate/pulse rate, respiratory rate and SpO2.
- Pulse oximetry: Measures oxygenation (SpO2) and pulse rate.
- Continuous Positive Airway Pressure (CPAP): Provides respiratory support to neonates with either upper airway obstruction or respiratory failure. CPAP delivers oxygen concentrations and distending airway pressures via the ventilator without the need for full endotracheal intubation, to help maintain adequate functional residual capacity within the alveoli and promote optimal gas exchange.
Assessment
A thorough respiratory assessment should be carried out on patients at commencement of shift, with cares, and when clinically indicated. Patients with PRS may be observed to have breathing movements but not actually have adequate air entry for adequate oxygenation and ventilation. This is why auscultation with a stethoscope is a vital component of respiratory assessment for PRS patients to ensure transmission of breaths.
Symptoms of respiratory compromise may not present immediately, and airway obstruction may be intermittent. Clinical signs may include, stridor, laboured breathing, apnoea, cyanosis, desaturations, bradycardia, irritability, and restlessness. Additional signs and symptoms may include difficulty feeding and poor weight gain. Poor weight gain may be an indication of chronic airway obstruction as the infants energy expenditure is increased secondary to airway obstruction and greater work of breathing.
Indications for NPT Insertion
- Obstructive episodes noted by medical, nursing, or allied health staff
- Overnight oximetry indicative of hypoxic episodes
- Sleep study indicative of obstructive episodes
- Poor feeding tolerance and insufficient weight gain
- Significant respiratory distress, further evidenced by hypercapnia on blood gas results
- Poor transmission of breaths and air entry on auscultation
Measurement of NPT Size
The optimal efficiency of a nasopharyngeal airway to maintain airway patency is significantly dependent on both internal diameter of the airway and the position of the distal tip. Selection of the size (diameter) of the ETT and length to place it at the nostril is outlined in the table below. This table should be utilized as a guide, and a thorough respiratory assessment should be conducted during insertion to ensure that the NPT is sitting at a length that bypasses the obstructing tongue. An estimate length can also be obtained by measuring from the tip if the nose to the ear tragus, then adding an additional 0.5cm.
The ideal position of an NPT should not extended past the epiglottis, and should sit approximately 1cm above. If the airway is sized too short, it may fail to alleviate obstruction, and if the length is too long it can stimulate and aggravate the cough and gag reflex.
Table 1.
|
Weight of neonate (kg)
|
Length of ETT (cm)
|
Diameter of ETT (mm)
|
|
< 2.5
|
7.0
|
3.0
|
|
2.5 - 3.5
|
7.5
|
3.5
|
|
> 3.5
|
8.0
|
4.0
|
Management
Modification of NPT
Equipment Required:
- ETT of appropriate size
- Comfeel
- Brown Leukoplast rigid tape, size 25mm
- Scissors
- Lubricant
- Nasogastric tube (NGT) (if required for feeding)
- Clear, occlusive dressing (e.g. Tegaderm, Opsite)
Procedure:
| 1. Open appropriate size ETT as guided by the above chart |

|
| 2. Cut ETT 5cm longer than the length it is measured to sit at the nostril (Eg – If inserting to 8cm, cut ETT at 13cm). Discard the end with adaptor attached |
 |
| 3. Cut along either side of the blue line (as close as possible to line) marked on the ETT so you are left with a blue line strand |

|
| 4. Cut along the right hand side of the number markings on the ETT |
 |
| 5. You should end up with 3 strands |
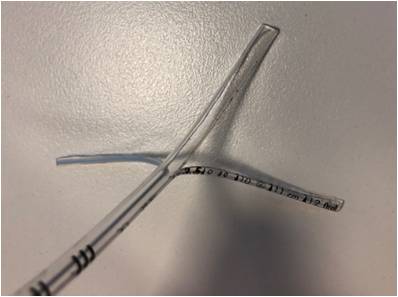
|
| 6. Cut the clear plastic strand in half to end up with 4 strands |

|
7. Cut off the clear plastic strand opposite the blue strip. You should be left with 3 strands ready for insertion
Note: Trim down the blue line strand to secure to the nasal bridge
|
 |
Insertion and Securing of NPT
A member of the medical team or senior member of nursing staff (if approved by medical team) should insert the initial NPT, and nurses may insert subsequent NPTs as required. A lateral neck x-ray should be obtained post insertion to confirm the position of the initial NPT. The position should be adjusted if necessary
according to the x-ray. If respiratory distress persists despite an appropriately positioned NPT, Continuous Positive Airway Pressure (CPAP) or endotracheal intubation should be considered. Please note when administering CPAP, single prong mode must be utilised, and the length should be the same length as the
NPT so that the level of tongue base
obstruction is bypassed.
Following successful insertion of an NPT, this should be documented in EMR as an LDA. Any new NPT insertions should be documented as a new LDA, and previous NPT’s removed.
Any patient with an NPT insitu should also have a laminated bedside card, which clearly states the size, length, and date the NPT was last changed.
Ensure that two health practitioners (one to insert the NPT, the other to provide comfort measures to the neonate) are present for the insertion of the NPT, and that the medical team/AUM is aware that the procedure is occurring. The initial NPT should
only be inserted after a thorough assessment of the neonate by a member of the medical team, and discussion with the consultant.
Prior to insertion, administer sucrose as per RCH nursing clinical guideline (
Sucrose oral for procedural pain management in infants), gather all required equipment, and perform hand hygiene.
1. There are two strands that will be taped to the face, and the thin blue strand will be trimmed and taped to the nose (see picture below). Please ensure the blue strand is trimmed and secured to the patient nose (see bottom picture), and not their forehead.
This helps to minimise movement and stabilise the NPT
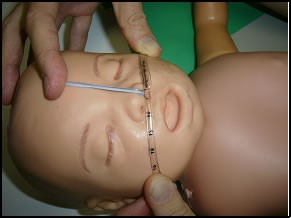
2. Apply comfeel to each cheek and the nose to preserve skin integrity, before insertion and taping.
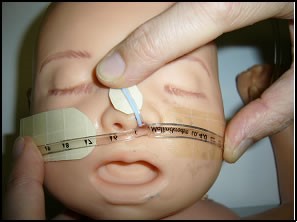
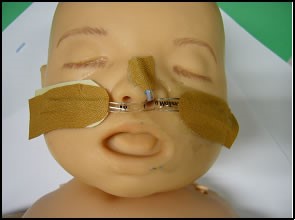
3. Apply lubricant to the tip of the NPT. Insert the tube into the nostril, and once passed through the choana, continue insertion slowly and carefully, until misting is seen in the tube, which will occur on exhalation. Secure the tube by placing the tape directly
over the straps on the cheeks and nose. Apply the tape as close to the nare as possible to prevent mobility of the tube after securing. Make sure that the nares are still pink, and monitor for any signs of pressure areas.
4. If insertion of a NGT is required, insert this into the other nostril, and secure onto the Leukoplast tape with Tegaderm/Opsite.
5. Ensure a lateral neck x-ray (with patient's head in neutral position) is taken after initial NPT insertion. If alteration is required, re-cut a new NPT according to the modified length required from the x-ray. Refer to x-ray below which shows a patient with an NPT in situ. The NPT
is sitting just above the epiglottis and below the posterior tongue, therefore relieving the obstruction.
.JPG)
Timing of NPT Changes
In the first 10 days post insertion of the NPT, it should be changed every 2-4 days or PRN if secretions are affecting tube patency. More frequent occlusions may occur during this time from the trauma of initial insertion. After this period it should be routinely changed every 5-7 days, with alternating nostrils utilised. If the NPT is required over long-term, size and length may need adjusting according to patient's growth. Further lateral neck x-rays are only required if clinically indicated.
Other indications to change tube:
- Frequent occlusions
- Obstructive episodes – Poor transmission of breaths, desaturations, bradycardia, cyanosis
- Blocked tube – Unable to pass suction catheter, clinical indications such as obstructive breathing, increased work of breathing, poor air entry, desaturation, bradycardia
- Query blocked tube – Resistance passing suction catheter
- Signs of pressure areas
Ensure spare modified NPT of same size, and one size smaller, with tapes pre-cut are available at the bedside at all times in case of accidental removal.
Skin Integrity
- Monitor the patient's skin integrity, inspecting the nares at least once per shift or more frequently, ensuring they are pink in appearance. If the nares are white, the tube needs to be re-taped to reduce pressure on the nostril.
- Pressure areas can occur if an NPT is incorrectly sized, too mobile, or tapes are secured too tightly across the cheeks and nares.
- If there is evidence of skin breakdown, medical team should be notified and NPT should be changed to the opposite nare.
- Tapes should be changed as required, if security of tube or patient skin integrity is becoming compromised, or with a routine NPT change.
- Ensure comfeel is always placed on the patient's cheeks to protect skin from damage caused by adhesive tape securing the NPT.
- Document any pressure areas on EMR and upload clinical picture for patient file.
Ongoing Assessment and Monitoring
A thorough respiratory assessment is necessary at shift commencement, and if patient's condition changes. Respiratory assessment should always include auscultation of patient's chest with a stethoscope to assess for air entry with breathing movements.
The presence of stridor indicates obstructed breathing, however absence of stridor does not indicate a patent airway.
Continuous pulse oximetry monitoring must be in place for all infants with an NPT inserted for upper airway obstruction. Cardio-respiratory monitoring is important in the first 10 days post insertion of an NPT, but if obstructive episodes have ceased, continuous pulse oximetry may be sufficient after discussion with medical team. If obstructive episodes are still occurring, or the NPT is removed for a trial period, continue with full cardio-respiratory monitoring in addition to the continuous pulse oximetry.
Continuous close observation for respiratory distress and obstructive episodes is essential, as respiratory monitoring may pick up breathing movements, but cannot measure adequate airway airflow.
Suction should be carried out prn and should never exceed more than 0.5cm beyond the tip of the NPT. Lavage by instillation of normal saline is not supported due to the risk of aspiration. If the NPT is suspected to be blocked, the tube will need to be changed.
NPT Trial Out
- NPT trial out occurs two weeks post initial insertion
- Prior to removing the NPT, ensure that the medical team and AUM are aware
- Have 2 x pre-cut NPT’s at the bedside ready to reinsert in case of airway obstruction -Have an NPT of the same size that was previously inserted, and also one size smaller
- Ensure all safety equipment including Neopuff and Suction are set up and readily available if required
- When the NPT is removed, the patient should be nursed supine unless otherwise ordered by the medical team
- If there are any concerns or signs of airway obstruction while the patient is supine, the medical team should be notified immediately for review
- If the patient shows signs of airway obstruction and requires reinsertion of an NPT, another trial out will occur 2 weeks post reinsertion
- After two failed attempts, the patient will proceed with surgical intervention
Other considerations
- Should an aerosol generating procedure be undertaken on a patient under droplet precautions then increase to airborne precautions by donning N95/P2 mask for at least the duration of the procedure.
- Family Centred Care - It is the responsibility of the clinicians caring for the infant requiring a modified NPT for PRS, to ensure that the parents understand the rationale for this treatment.
- The bedside nurse should encourage and engage the family as much as possible to attend to their babies cares, including bathing, feeding, and skin to skin contact. The nurse should also encourage the parents to view the COCOON webpage. COCOON is a model of care utilised on the
Butterfly ward that aims to improve health outcomes for babies, and to improve the experience of families who have babies on the ward.
- Special Considerations - An NPT is not suitable for use in all infants with PRS. Individual assessment of the type and level of obstruction is required.
- Medical
staff must insert the modified NPT if it is used in any condition other than
Pierre Robin Sequence
- There is still a risk that airway obstruction can occur despite a nasopharyngeal tube in place
Companion Documents
Links
Evidence Table
The evidence table for Nasopharyngeal Tube (NPT) modified for Pierre Robin Sequence (PRS) can be viewed here.
Please remember to
read the disclaimer.
The development of this nursing guideline was coordinated by Emma Lewis, Clinical Support Nurse, Butterfly, and approved by the Nursing Clinical Effectiveness Committee. Updated August 2020.